Immunodetection made easy, from identifying HLA-antibody interactions to staining and isolating immune cell populations
HLA Class I Proteins
HLA Protein Technologies Inc’s functionally intact HLA-Class I proteins have been technologically modified and are now available in soluble form without interfering transmembrane domains. Select from a wide range of alleles that properly present antigenic regions to study antibody or cellular interactions.

Choose from a range of HLA alleles
HLA-A
Choose from more than 35 HLA-A alleles to detect, profile, or monitor antigen-specific immune responses from antibodies to immune cell populations
HLA-B
Select from a pool of over 65 HLA-B alleles to drive your immunological research to identify antibody immune responses or visualize antigen-specific immune cells
HLA-C
Discover a selection of over 20 HLA-C alleles, the dominant ligand for KIR on NK cells for your immunological research
HLA-G
Explore our HLA-G portfolio—designed for true-to-biology structure, verified function, and cross-disciplinary immunological discovery.
Now Available!
HLA Class I Basics
Soluble HLA (sHLA) molecules are recombinant, endogenously loaded and naturally folded glycoproteins. Using proprietary technology, HLA Protein Technologies Inc truncates these proteins that are normally attached to the surface of the cell membrane, just before the trans-membrane and cytoplasmic domain, allowing them to be secreted and easily purified without associated cell membranes.
Structurally, sHLA Class I proteins consist of a heavy chain (comprised of a1, a2, and a3 domains), a light chain (b2m) and the peptide they present forming a trimeric complex. Various sHLA alleles have an added purification tag (VLDL) at the carboxy end of the α3 domain.
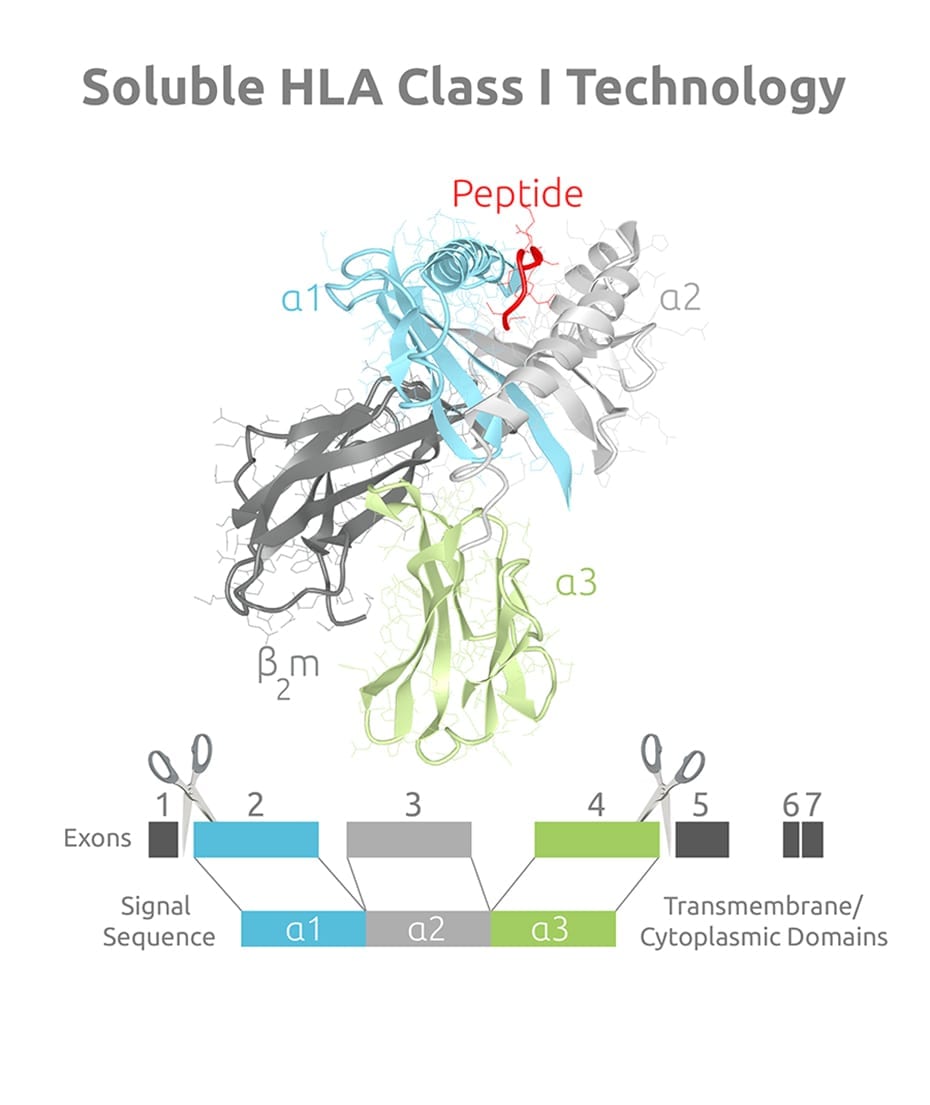

Protein Characteristics
HLA Class I genes are highly polymorphic, representing the genetically most variable gene region by encoding thousands of different alleles. HLA Protein Technologies Inc offers a wide selection of single-specificity HLA proteins with broad population coverage. Since sHLA molecules lack the transmembrane portion, these proteins are not retained on the cell surface but readily secreted allowing economic production and purification thereby eliminating the inherent problems of detergent cell lysates.
Soluble Class I proteins are well characterized, proven to present native, antigenic epitopes recognized by specific antibodies, demonstrate high stability and integrity of their structures over time and are compliable on many application platforms as well as adaptable for individual needs.
You might also be interested in...
Soluble HLA Class II Proteins
Mammalian produced, zipper stabilized, endogenously loaded, and naturally folded recombinant soluble HLA Class II proteins
T Cell Epitope Mapping
Discover your next immunotherapy targets utilizing a high-throughput approach to create individual epitope maps on any chosen protein that is potentially recognized by T cells.
Peptide Epitope Validation
Validate individual peptide epitope candidates by determining their physical binding characteristics and directly measure accurate IC50 values in order to judge and properly prioritize their immunogenic potential.

